 Department of Chemistry
Department of Chemistry
-
About
-
Research
-
Academics
-
People
-
Board
Seminar
Seminar
Bismuth Redox Catalysis
- POSTED DATE : 2022-09-22
- WRITER : 화학과
- HIT : 2529
- DATE : 2022년 9월 29일(목) 오후 4시 30분
- PLACE : Webex
세미나가 다음주 목요일(9월 29일) 오후 4시 30분에 개최됩니다.
* 이번 세미나는 온라인으로 진행되오니 착오없으시길 바랍니다. 온라인으로 많은 참여 부탁드립니다.
===============================================================================
제 목 : Bismuth Redox Catalysis
연 사 : Dr. Josep Cornella(Max-Planck-Institut für Kohlenforschung)
일 시 : 2022년 9월 29일(목) 오후 4시 30분
<Webex참여>
방번호: 170 974 2739
링크: https://skku-ict.webex.com/meet/chem
================================================================================
Bismuth Redox Catalysis
Josep Cornella
Max-Planck-Institut für Kohlenforschung, Mülheim an der Ruhr, 45470, Germany
cornella@kofo.mpg.de
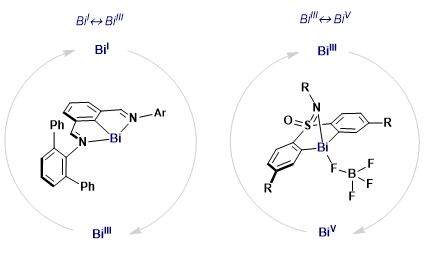
The ability of the main group element bismuth (Bi) to maneuver between different oxidation states in a catalytic redox cycle will be presented. We will show how Bi mimics the canonical organometallic steps of a transition metal, thus challenging the current dogmas of redox catalysis.[1] A series of Bi complexes capable of revolving between oxidation states Bi(I)/Bi(III) and Bi(III)/Bi(V) have been unlocked and applied in various contexts of catalysis for organic synthesis. For example, capitalizing on the Bi(III)/Bi(V) redox pair, we have developed a catalytic protocol for the C‒F[2] and C‒OTf [3] bond formation from aryl boronic esters. On the other hand, a low-valent redox manifold based on Bi(I)/Bi(III) enabled the reduction of hydrazines and nitro compounds[4], the catalytic decomposition of the rather inert nitrous oxide (N2O),[5] the catalytic hydrodefluorination of C(sp2)‒F bonds.[6] In addition, we will show how one-electron pathways are also accessible, thus providing a platform for SET processes capitalizing on the triad Bi(I)/Bi(II)/Bi(III) for organic synthesis.[7] Finally, we will also show how redox-neutral catalytic pathways can unlock novel organic transformations via canonical organometallic steps.[8] For all methodologies, a combination of rational ligand design with an in depth analysis of all the catalytic steps proved crucial to unfold the catalytic properties of such an intriguing element of the periodic table.
References
[1] Moon, H. –W. ; Cornella, J. ACS. Catal. 2022, 12, 1382.
[2] Planas, O.; Wang, F.; Leutzsch, M.; Cornella, J. Science, 2020, 367, 313..
[3] Planas, O.; Peciukenas, V.; Cornella, J. J. Am. Chem. Soc. 2020, 142, 11382
[4] Wang, F.; Planas, O.; Cornella, J. J. Am. Chem. Soc. 2019, 141, 4235.
[5] Pang. Y.; Leutzsch, M.; Nöthling, N.; Cornella, J. J. Am. Chem. Soc. 2020, 142, 19473.
[6] Pang, Y.; Leutzsch, M.; Nöthling, N.; Katzenburg, F.; Cornella, J. J. Am. Chem. Soc. 2021, 143, 12487.
[7] Mato, M. ; Spinnato, D. ; Cornella, J. unpublished results
[8] Magre, M. Cornella, J. J. Am. Chem. Soc. 2021, 143, 21497.
 발전기금
발전기금
 NEXT
NEXT PREV
PREV